Home > Press > Nature Communications: How metal clusters grow: Researchers from Marburg and Karlsruhe bring light into cluster formation; basis for customized optoelectronic and magnetic materials
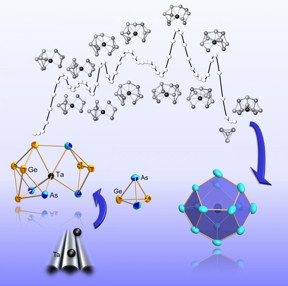 |
| Step by step towards the complete shell: formation of a metal cluster from the atomic constituents to the compound.
Photo: Dehnen Group, Philipps-Universit�t Marburg |
Abstract:
First the nucleus, then the shell: Researchers from Marburg and Karlsruhe have studied stepwise formation of metal clusters, smallest fractions of metals in molecular form. The shell gradually forms around the inner atom rather than by later inclusion of the central atom. Knowledge of all development steps may allow for customized optoelectronic and magnetic properties, as is reported by the researchers in the science journal Nature Communications. (DOI: 10.1038/NCOMMS10480)
Nature Communications: How metal clusters grow: Researchers from Marburg and Karlsruhe bring light into cluster formation; basis for customized optoelectronic and magnetic materials
Karlsruhe, Germany | Posted on February 29th, 2016To specifically synthesize chemical compounds, mechanisms responsible for their formation have to be known. "Purely inorganic compounds are a black box in this respect," Florian Weigend of Karlsruhe Institute of Technology (KIT) and Stefanie Dehnen of Philipps-Universit�t Marburg, the corresponding authors of this study, explain. "This particularly applies to the formation of multinucleated metal complexes, so-called clusters." Processes of transforming metal clusters take place rapidly. Normally, it is not possible to observe these processes and the intermediate products. "Even the very first steps are largely unknown and can only be identified by combining synthesis with measurement and computerized chemical modeling," the experts say. If all development steps were known, metal clusters with finely adjusted optoelectronic and magnetic properties might be customized for technical applications.
In the study published now, the team reports the formation of a multinucleated metal cluster by first synthesizing a series of variably sized clusters of the metalloids germanium and arsenic. Larger clusters have an atom of the transition metal tantalum in the center of the cage molecules. Measurements and computer simulations suggest that the transition metal very early plays a role in cluster formation. "It may be considered a type of catalyst that initiates the formation and breaking of bonds," Weigend and Dehnen point out. Altogether, the findings show that the transition metal does not settle into an earlier formed cluster shell, but that the shell of the cluster gradually forms around the atom in the center. "The results can be generalized for a whole family of metallic cluster compounds," Weigend and Dehnen say.
Apart from chemical syntheses and measurements in Marburg, extensive computer simulations with the largely KIT-developed quantum chemistry program package TURBOMOLE were made at KIT. In this way, the role of the many isomers in the reaction was determined. Isomers are chemical compounds of identical composition, but with different spatial arrangements of the atoms. "Thanks to the calculation of reaction paths, we have found that transformations of isomers are possible with a relatively small energy expenditure," Weigend says.
Apart from Dehnen and Weigend and doctoral student Stefan Mit-zinger, the Humboldt scholarship holder Lies Broeckaert and Werner Masse, former Head of the Central Laboratory for Crystal Structure Analysis, are involved in the present publication. Underlying research was supported by the Alexander von Humboldt Foundation, Friedrich Ebert Foundation, and the German Research Foundation.
####
About Karlsruhe Institute of Technology
Karlsruhe Institute of Technology (KIT) pools its three core tasks of research, higher education, and innovation in a mission. With about 9,400 employees and 24,500 students, KIT is one of the big institutions of research and higher education in natural sciences and engineering in Europe.
KIT - The Research University in the Helmholtz Association
Since 2010, the KIT has been certified as a family-friendly university.
For more information, please click here
Contacts:
Monika Landgraf
49-721-608-47414
For further information, please contact:
Kosta Schinarakis, PKM
Science Scout
Phone: +49 721 608 41956
Fax: +49 721 608 43658
Copyright © Karlsruhe Institute of Technology
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
![]() Website of the Weigend Research Group: (in German only):
Website of the Weigend Research Group: (in German only):
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Chemistry
![]() Projecting light to dispense liquids: A new route to ultra-precise microdroplets January 30th, 2026
Projecting light to dispense liquids: A new route to ultra-precise microdroplets January 30th, 2026
![]() From sensors to smart systems: the rise of AI-driven photonic noses January 30th, 2026
From sensors to smart systems: the rise of AI-driven photonic noses January 30th, 2026
![]() "Nanoreactor" cage uses visible light for catalytic and ultra-selective cross-cycloadditions October 3rd, 2025
"Nanoreactor" cage uses visible light for catalytic and ultra-selective cross-cycloadditions October 3rd, 2025
![]() Cambridge chemists discover simple way to build bigger molecules � one carbon at a time June 6th, 2025
Cambridge chemists discover simple way to build bigger molecules � one carbon at a time June 6th, 2025
Govt.-Legislation/Regulation/Funding/Policy
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
![]() New imaging approach transforms study of bacterial biofilms August 8th, 2025
New imaging approach transforms study of bacterial biofilms August 8th, 2025
Materials/Metamaterials/Magnetoresistance
![]() First real-time observation of two-dimensional melting process: Researchers at Mainz University unveil new insights into magnetic vortex structures August 8th, 2025
First real-time observation of two-dimensional melting process: Researchers at Mainz University unveil new insights into magnetic vortex structures August 8th, 2025
![]() Researchers unveil a groundbreaking clay-based solution to capture carbon dioxide and combat climate change June 6th, 2025
Researchers unveil a groundbreaking clay-based solution to capture carbon dioxide and combat climate change June 6th, 2025
![]() A 1960s idea inspires NBI researchers to study hitherto inaccessible quantum states June 6th, 2025
A 1960s idea inspires NBI researchers to study hitherto inaccessible quantum states June 6th, 2025
![]() Institute for Nanoscience hosts annual proposal planning meeting May 16th, 2025
Institute for Nanoscience hosts annual proposal planning meeting May 16th, 2025
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Interviews/Book Reviews/Essays/Reports/Podcasts/Journals/White papers/Posters
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








