Home > Press > Silver cycle: New evidence for natural synthesis of silver nanoparticles
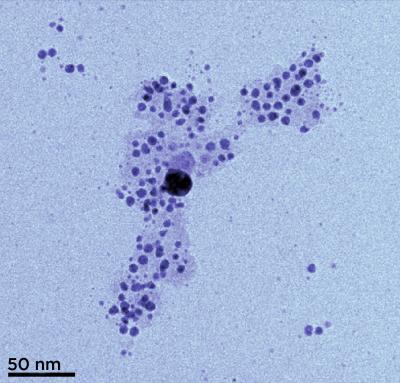 |
| Transmission electron microscopy (TEM) image of silver nanoparticles formed from silver ions in solution with humic acid. The acid tends to coat the nano particles (visible here as a pale cloud), keeping them in a colloidal suspension instead of clumping together. (Color added for clarity.)
Credit: SUNY, Buffalo |
Abstract:
Nanoparticles of silver are being found increasingly in the environment—and in environmental science laboratories. Because they have a variety of useful properties, especially as antibacterial and antifungal agents, silver nanoparticles increasingly are being used in a wide variety of industrial and consumer products. This, in turn, has raised concerns about what happens to them once released into the environment. Now a new research paper* adds an additional wrinkle: Nature may be making silver nanoparticles on its own.
Silver cycle: New evidence for natural synthesis of silver nanoparticles
Gaithersburg, MD | Posted on May 11th, 2011A team of researchers from the Florida Institute of Technology (FIT), the State University of New York (SUNY), Buffalo, and the National Institute of Standards and Technology (NIST) reports that, given a source of silver ions, naturally occurring humic acid will synthesize stable silver nanoparticles.
"Our colleague, Virender Sharma, had read an article in which they were using wine to form nanoparticles. He thought that, based on the similar chemistry, we should be able to produce silver nanoparticles with humic acids," explains FIT chemist Mary Sohn. "First we formed them by traditional methods and then we tried one of our river sediment humic acids. We were really excited that we could see the characteristic yellow color of the nanoparticles." Samples were sent to Sarbajit Banerjee at SUNY Buffalo and Robert MacCuspie at NIST for detailed analyses to confirm the presence of silver nanoparticles.
"Humic acid" is a complex mixture of many organic acids that are formed during the decay of dead organic matter. Although the exact composition varies from place to place and season to season, humic acid is ubiquitous in the environment. Metallic nanoparticles, MacCuspie explains, have characteristic colors that are a direct consequence of their size.** Silver nanoparticles appear a yellowish brown.
The team mixed silver ions with humic acid from a variety of sources at different temperatures and concentrations and found that acids from river water or sediments would form detectable silver nanoparticles at room temperature in as little as two to four days. Moreover, MacCuspie says, the humic acid appears to stabilize the nanoparticles by coating them and preventing the nanoparticles from clumping together into a larger mass of silver. "We believe it's actually a similar process to how nanoparticles are synthesized in the laboratory," he says, except that the lab process typically uses citric acid at elevated temperatures.
"This caught us by surprise because a lot of our work is focused on how silver nanoparticles may dissolve when they're released into the environment and release silver ions," MacCuspie says. Many biologists believe the toxicity of silver nanoparticles, the reason for their use as an antibacterial or antifungal agent, is due to their high surface area that makes them an efficient source of silver ions, he says, but "this creates the idea that there may be some sort of natural cycle returning some of the ions to nanoparticles." It also helps explain the discovery, over the past few years, of silver nanoparticles in locations like old mining regions that are not likely to have been exposed to man-made nanoparticles, but would have significant concentrations of silver ions.
* N. Akaighe, R.I. MacCuspie, D.A. Navarro, D.S. Aga, S. Banerjee, M. Sohn and V.K. Sharma. Humic acid-induced silver nanoparticle formation under environmentally relevant conditions. Environmental Science & Technology, Published online Apr. 1, 2011. dx.doi.org/10.1021/es103946g.
** The effect is called "surface plasmon resonance" and is caused by surface electrons across the nanoparticle oscillating in concert.
####
For more information, please click here
Contacts:
Michael Baum
301-975-2763
Copyright © National Institute of Standards and Technology (NIST)
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Laboratories
![]() Researchers develop molecular qubits that communicate at telecom frequencies October 3rd, 2025
Researchers develop molecular qubits that communicate at telecom frequencies October 3rd, 2025
![]() A battery’s hopping ions remember where they’ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery’s electrolyte is surprisingly complicated February 16th, 2024
A battery’s hopping ions remember where they’ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery’s electrolyte is surprisingly complicated February 16th, 2024
Govt.-Legislation/Regulation/Funding/Policy
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
![]() New imaging approach transforms study of bacterial biofilms August 8th, 2025
New imaging approach transforms study of bacterial biofilms August 8th, 2025
Discoveries
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse ‘quantum scrambling’ : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse ‘quantum scrambling’ : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Environment
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Researchers unveil a groundbreaking clay-based solution to capture carbon dioxide and combat climate change June 6th, 2025
Researchers unveil a groundbreaking clay-based solution to capture carbon dioxide and combat climate change June 6th, 2025
Safety-Nanoparticles/Risk management
![]() Tiny nanosheets, big leap: A new sensor detects ethanol at ultra-low levels January 30th, 2026
Tiny nanosheets, big leap: A new sensor detects ethanol at ultra-low levels January 30th, 2026
![]() Onion-like nanoparticles found in aircraft exhaust May 14th, 2025
Onion-like nanoparticles found in aircraft exhaust May 14th, 2025
![]() Closing the gaps — MXene-coating filters can enhance performance and reusability February 28th, 2025
Closing the gaps — MXene-coating filters can enhance performance and reusability February 28th, 2025
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








