Home > Press > Surprise shift: A first-of-a-kind switch in chemical bonding by a zirconium atom spotted by scientists
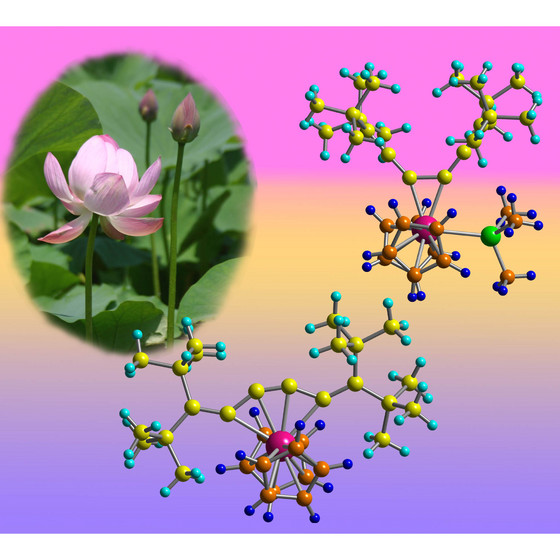 |
| Figure 1: Schematic showing that adding or removing an extra chemical group (green) to the zirconium atom (pink) can open and close the structure of the molecule like the petals of a lotus flower. |
Abstract:
A zirconium atom can switch easily between two different bonding patterns in an unusual molecule created by Japanese scientists.
Surprise shift: A first-of-a-kind switch in chemical bonding by a zirconium atom spotted by scientists
Japan | Posted on April 12th, 2009The molecule's odd behavior is the first example of this particular type of chemical bonding shift, according to Noriyuki Suzuki of RIKEN's Advanced Science Institute in Wako, and his colleagues at Saitama University and Saitama Institute of Technology. "These complexes have very unique structures and show interesting movement," says Suzuki. An example is shown in Figure 1.
The team discovered the phenomenon when they were experimenting with a molecule (hexapentaene) made from a chain of six carbon atoms, all doubly bonded to each other. The chain is surrounded by a long cloud of delocalized electrons, which can bond with a zirconium-based compound to create a new complex.
Once the new complex has formed, the zirconium atom normally tends to bridge between carbon atoms in different parts of the chain, creating a five-membered ring. But when very bulky groups were added to each end of the chain, the zirconium switches its allegiance so that it sticks to just the central carbon-carbon double bond.
The complex could be toggled between its two bonding modes by adding or removing other chemical groups such as phosphines around the zirconium atom. Further experiments showed that this sort of shift was the first step in a reaction the scientists had previously studied, where adding an isocyanide chemical to the zirconium complex created a compound with a small ring of four carbon atoms at its heart.
This switching behavior is well known in certain ring-shaped organic molecules, but is much rarer in these molecular chains, and is unprecedented with this particular compound. "It suggests the possibility of a molecular motion like scissors or tongs," says Suzuki. The research is published in the Journal of the American Chemical Society1.
Suzuki's team has spent several years investigating a range of such zirconium complexes, which were long assumed to be too unstable to isolate2,3.
Although there are no immediate applications for this family of complexes, Suzuki suggests that it may be possible to use the scissoring action identified in their latest research to capture another molecule or ion. "We might be able to achieve a molecular machine that catches a certain target," says Suzuki.
Reference
1. Suzuki, N., Hashizume, D., Yoshida, H., Tezuka, M., Ida, K., Nagashima, S. & Chihara, T. Reversible haptotropic shift in zirconocene-hexapentaene complexes. Journal of the American Chemical Society 131, 2050-2051(2009) .
2. Suzuki, N., Nishiura, M. & Wakatsuki, Y. Isolation and structural characterization of 1-zirconacyclopent-3-yne, five-membered cyclic alkynes. Science 295, 660-663 (2002).
3. Suzuki, N., Hashizume, D., Koshino, H. & Chihara, T. Transformation of a 1-zirconacyclopent-3-yne, a five-membered cycloalkyne, into a 1-zirconacyclopent-3-ene and formal "1-zirconacyclopenta-2,3-dienes". Angewandte Chemie International Edition 47, 5198-5202 (2008).
The corresponding author for this highlight is based at the RIKEN Chemical Analysis Team
####
For more information, please click here
Copyright © Riken
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Chemistry
![]() Projecting light to dispense liquids: A new route to ultra-precise microdroplets January 30th, 2026
Projecting light to dispense liquids: A new route to ultra-precise microdroplets January 30th, 2026
![]() From sensors to smart systems: the rise of AI-driven photonic noses January 30th, 2026
From sensors to smart systems: the rise of AI-driven photonic noses January 30th, 2026
![]() "Nanoreactor" cage uses visible light for catalytic and ultra-selective cross-cycloadditions October 3rd, 2025
"Nanoreactor" cage uses visible light for catalytic and ultra-selective cross-cycloadditions October 3rd, 2025
![]() Cambridge chemists discover simple way to build bigger molecules � one carbon at a time June 6th, 2025
Cambridge chemists discover simple way to build bigger molecules � one carbon at a time June 6th, 2025
Discoveries
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








